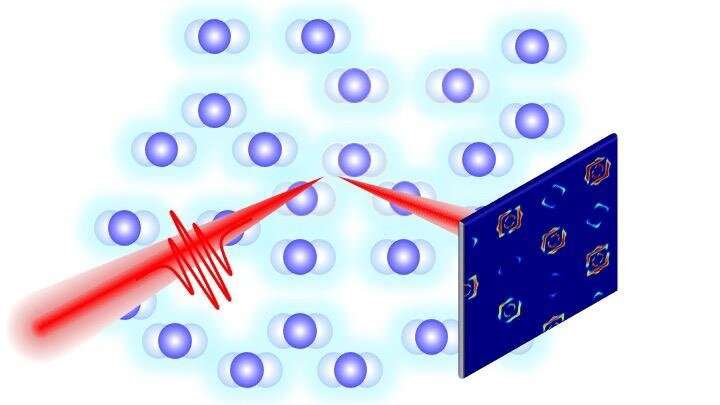
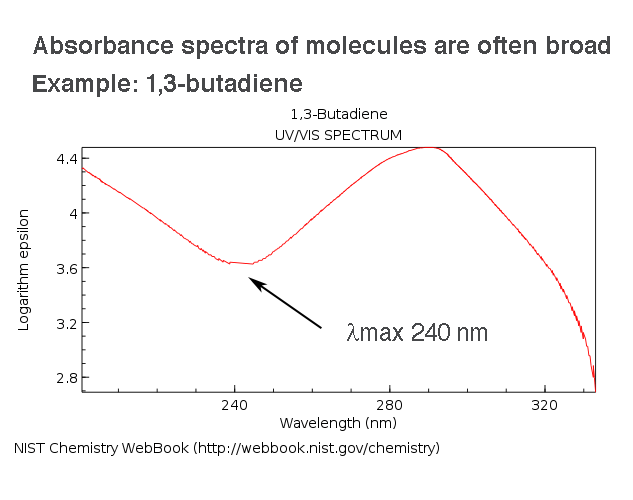
Coupling between Harmonic Vibrations Influences Quantum Beating Signatures in Two-Dimensional Electronic Spectra | The Journal of Physical Chemistry C

PDF) Shape and size of electron are determine by the amount of photon energy incidence on it 5e7f7e40d7564 (1) | Saddam H U S A I N Dhobi - Academia.edu

Electron–vibration coupling induced renormalization in the photoemission spectrum of diamondoids | Nature Communications

Question Video: Calculating the Kinetic Energy of an Electron Moving through a Negative Potential Difference | Nagwa
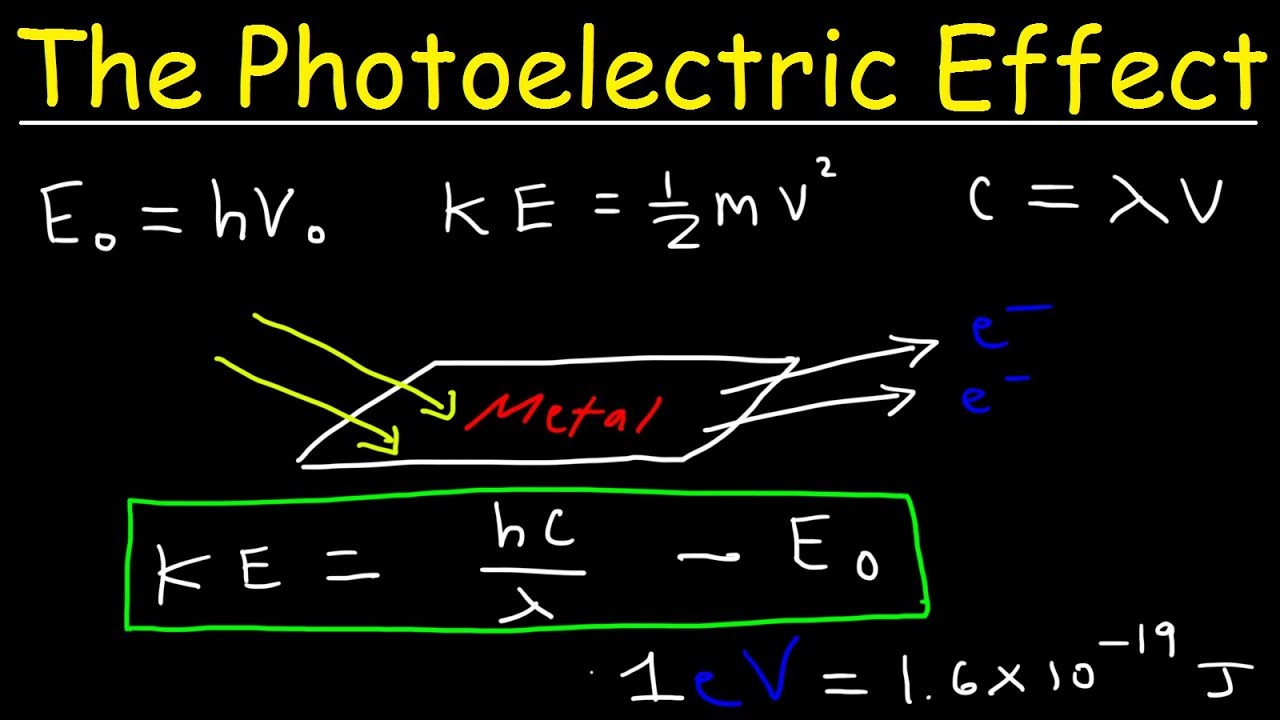
Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr - YouTube

About these slides These slides are used as part of my lessons and shouldn't be considered comprehensive There's no excuse for not turning up to lessons! - ppt video online download